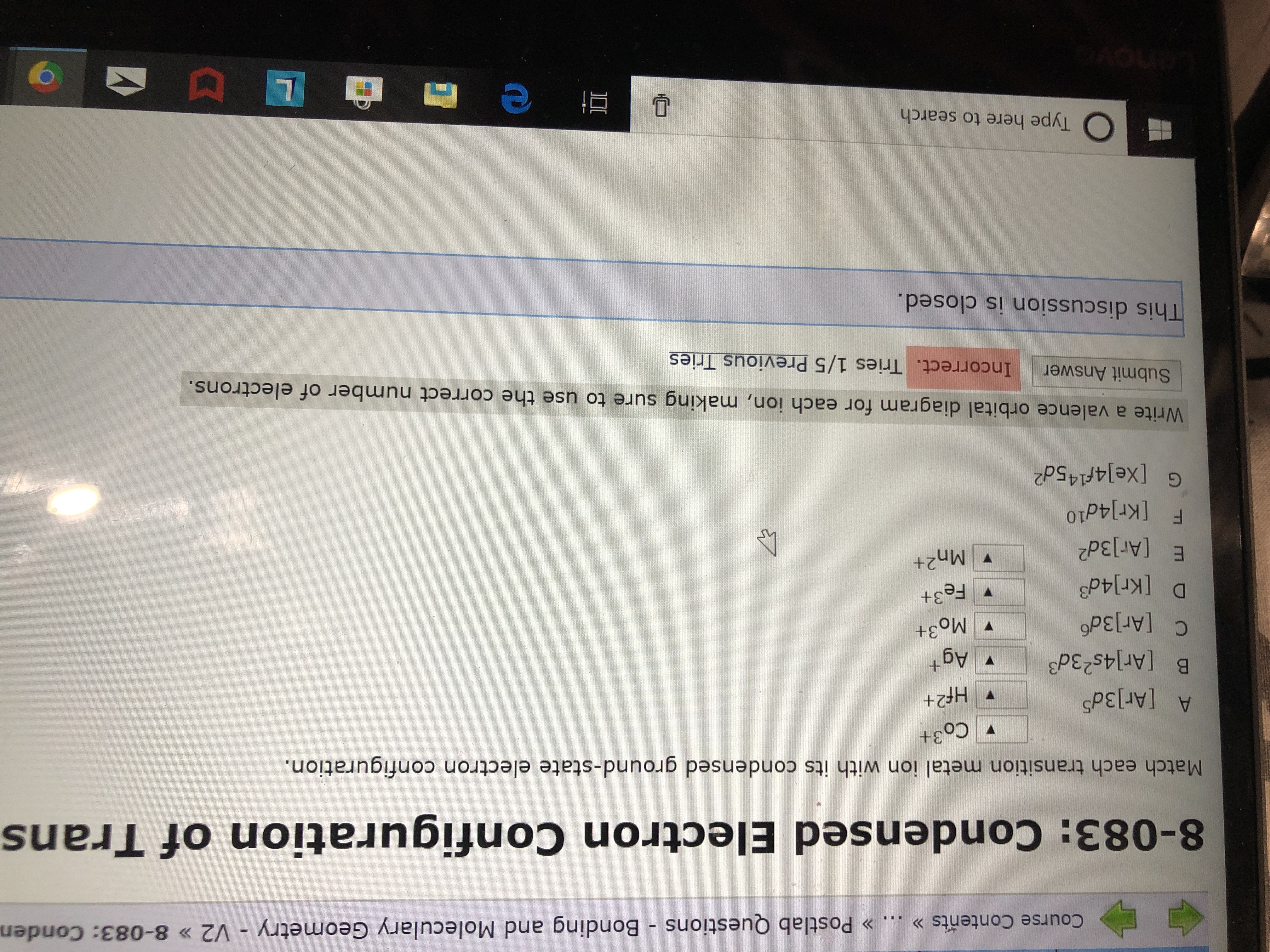
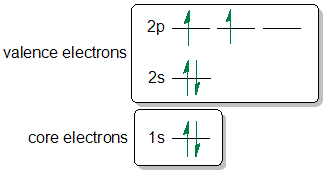
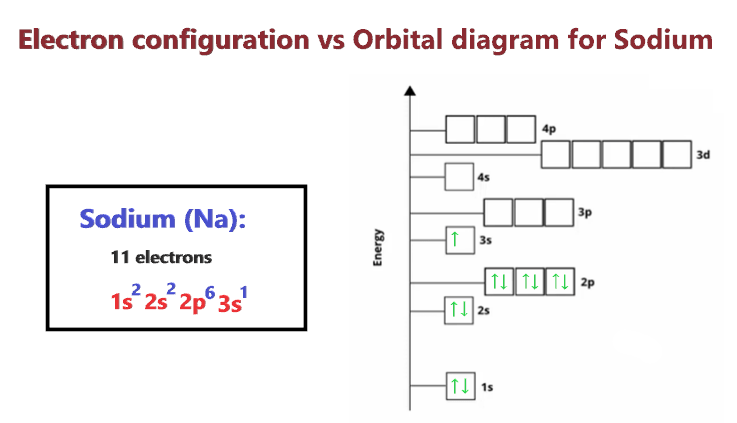
For each atom/ion, provide the condensed ground state electron configurations, the box diagram of the valence electrons, and the number of unpaired electrons. S, N, Cr, Cr3+, Fe2+, N3-, As | Homework.Study.com
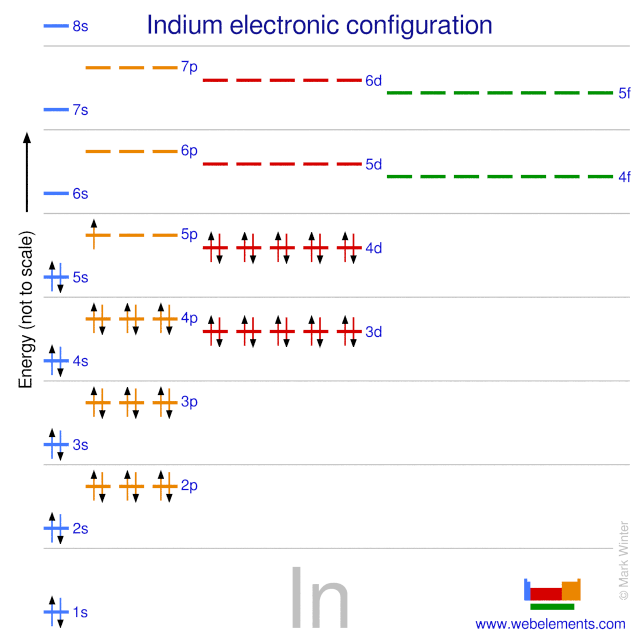
![Answered] Predict the ground-state electron configuration of the following ions. Write the answer in - Brainly.com Answered] Predict the ground-state electron configuration of the following ions. Write the answer in - Brainly.com](https://us-static.z-dn.net/files/db8/9ccbe99b18b11817c99d88b41e80bb7f.png)
Answered] Predict the ground-state electron configuration of the following ions. Write the answer in - Brainly.com

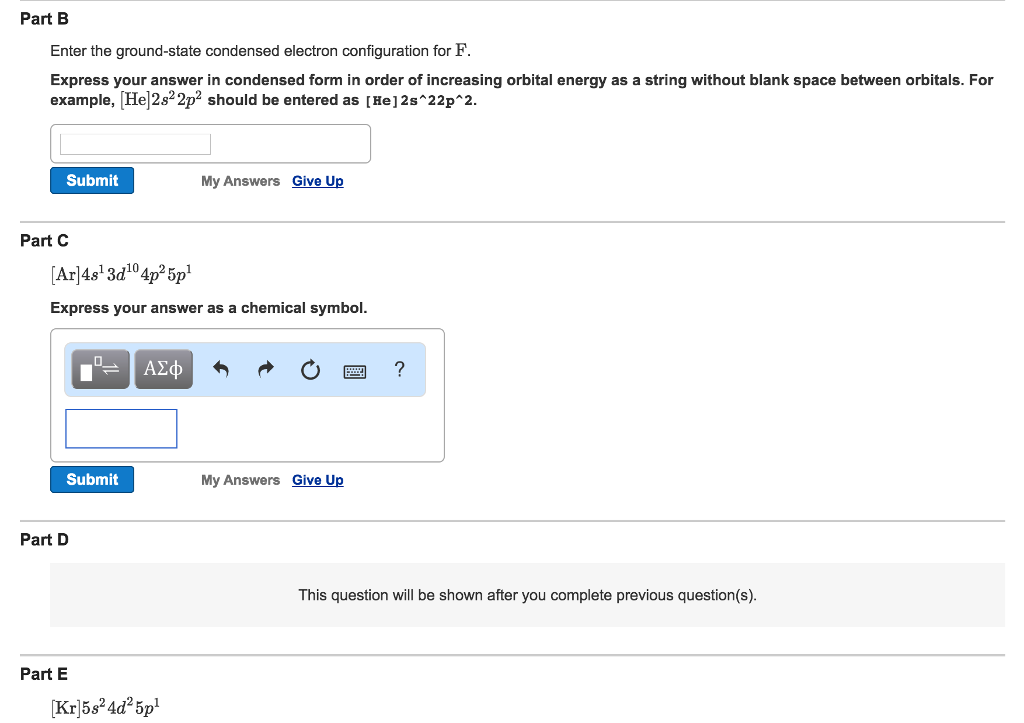
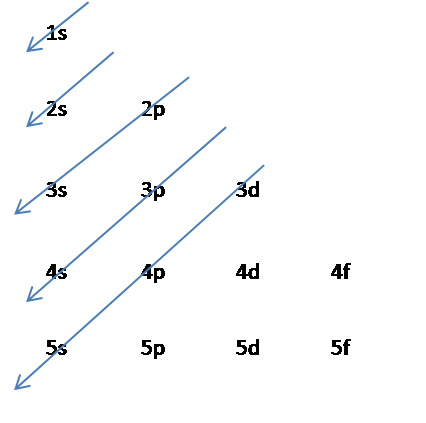
SOLVED:Write the condensed ground-state electron configuration for each of the following elements. The element's atomic number is given in parentheses. (a) \mathrm{Li}(3) (b) \mathrm{Ne}(10) (c) Be (4) (d) C(6) (e) \operatorname{Mg}(12)

SOLVED:Write the condensed ground-state electron configuration for each of the following elements. The element's atomic number is given in parentheses. (a) \mathrm{Li}(3) (b) \mathrm{Ne}(10) (c) Be (4) (d) C(6) (e) \operatorname{Mg}(12)